 
Scholarly yet accessible to those with basic physics knowledge, this book should be of interest to historians of science, professional scientists and engineers, teachers and students of physics, and general readers interested in the development of modern physics. 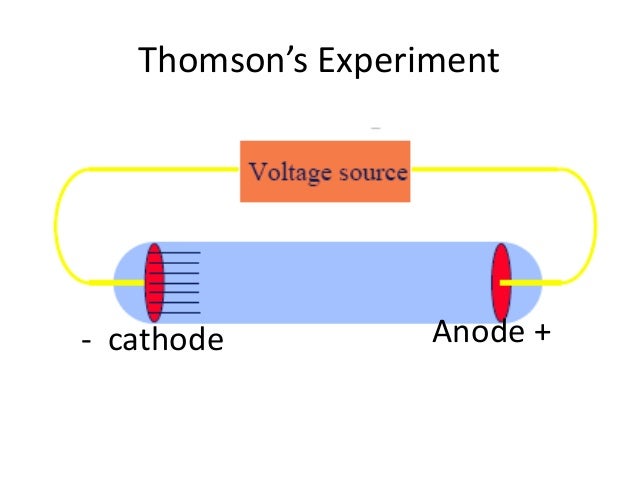
The author also focuses on the controversies over N-rays, Becquerel's positive electron, and the famous Ehrenhaft-Millikan dispute over subelectrons. It explores the events leading to Millikan's unambiguous isolation of the electron and the simultaneous circumstances surrounding the birth of Ernest Rutherford's nuclear atom and the discovery of radioactivity in 1896. It also provides an overview of the history of modern physics up to the advent of the old quantum theory around 1920.Ĭonsolidating scholarly material while incorporating new material discovered by the well-respected author, the book covers the continental and English race for the source of the cathode rays, culminating in Thomson's corpuscle in 1897. The book traces the evolution of the concept of electrical charge, from the earliest glow discharge studies to the final cathode ray and oil drop experiments of J.J. Thomson's Electron presents the compelling story of the discovery of the electron and its role as the first subatomic particle in nature. Flash of the Cathode Rays: A History of J.J. Thomson measured the deflection of the beam using a ruler etched on the end of. You can see a simulation of this glow on the far right of the applet diagram, as shown in Figure 2. Thomson was able to apply electric and magnetic fields to manipulate the rays, which eventually convinced the physics world that they were composed of tiny particles, electrons, opposed to waves in the now-rejected ether.įind out more about Thomson and the story of the first subatomic particle here, or visit the Museum to see Thomson’s cathode-ray tube in the Collider exhibition. If you’re interested in the details of how Thomson and Everett conducted their experiments visit the Cavendish Lab’s outreach page here.The electron is fundamental to almost all aspects of modern life, controlling the behavior of atoms and how they bind together to form gases, liquids, and solids. Thomson managed to estimate its magnitude by performing experiments with charged particles in gases. In Thomson’s experiment, a fluorescent material was coated on the end of the tube to produce a glowing dot where the cathode rays hit. Only when almost all the air has been removed to create a high vacuum – a state that would shatter ordinary glass vessels – can the rays travel the full length of the tube without bumping into air molecules. The quality of Everett’s glassblowing was absolutely crucial for the experiments to work.Ĭathode-rays are produced when an electric current is passed through a vacuum tube. Since the atom was neutral, Thomson suggested that the negatively charged electron equaled the positively charged proton and neutrons had no charges. Everett made all of Thomson’s apparatus, and was responsible for operating it – in fact, he generally forbade Thomson from touching anything delicate on the grounds that he was “exceptionally helpless with his hands”. Cambridge’s Cavendish Laboratory, where Thomson spent his scientific career, also has an original tube in its collection.Įach tube was custom-made by Thomson’s talented assistant, Ebenezer Everett, a self-taught glassblower. 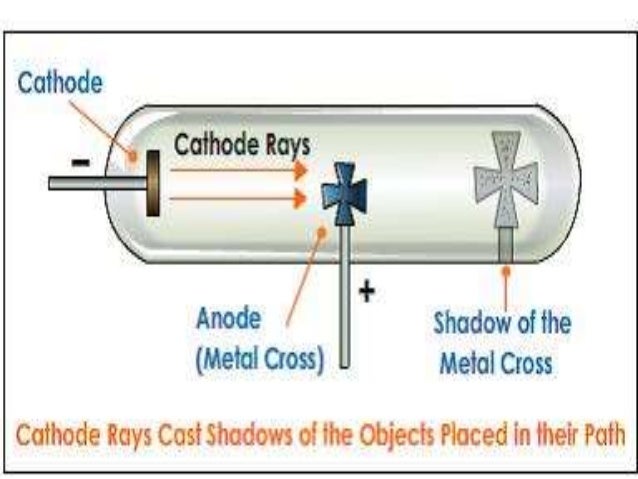
Using more than one cathode-ray tube in 1897 for his experiments, Thomson managed to identify a particle 1,000 times smaller than the then known smallest piece of matter: a hydrogen atom. I had read lots about Thomson’s famous experiments on the electron – the first subatomic particle to be discovered – but to actually see and touch his apparatus myself, to notice the blackened glass and the tube’s minute features that are omitted in books, brought the object to life. Holding the delicate glass cathode-ray tube in my hands, once used by the great physicist JJ Thomson, was an incredible treat, and an experience I will never forget. Rupert Cole celebrates JJ Thomson’s birthday with a look at one of the star objects in our Collider exhibition. He was well-known for the discovery of the electron. Rupert Cole celebrates JJ Thomson's birthday with a look at one of the star objects in our Collider exhibition. Sir Joseph John Thomson was a British physicist and Nobel Laureate. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
